If the phosphate purification bottleneck can be resolved, LFP batteries will play an important role in EVs and energy storage.
Iron phosphate and lithium-ion batteries
Lithium-ion batteries power a variety of devices, from smartphones and laptops to electric vehicles (EVs) and battery energy storage systems. One of the key components of a lithium-ion battery is the cathode material. Due to the need for high energy densities, cathodes made of oxides of either nickel, cobalt, manganese or aluminum are popular, especially for the long charge intervals they can provide to EVs. Recently, however, cathodes made from iron phosphate (LFP) have become increasingly popular, increasing the demand for phosphate production and purification.
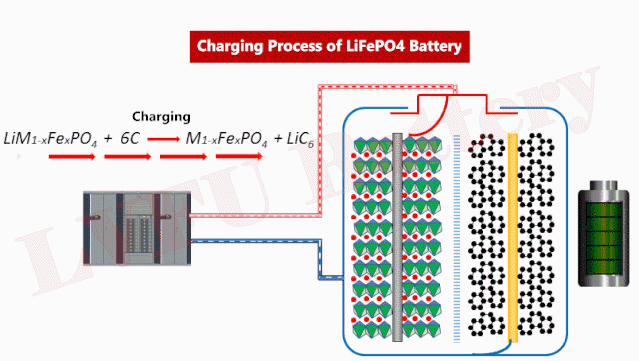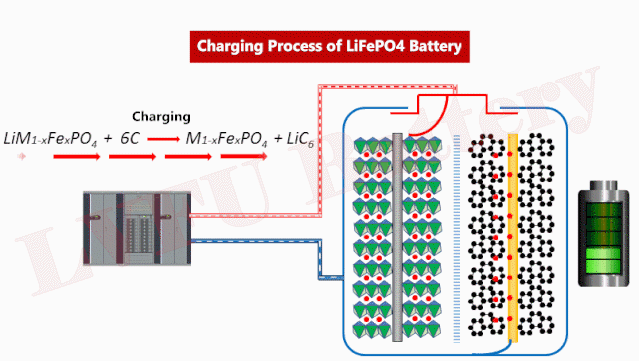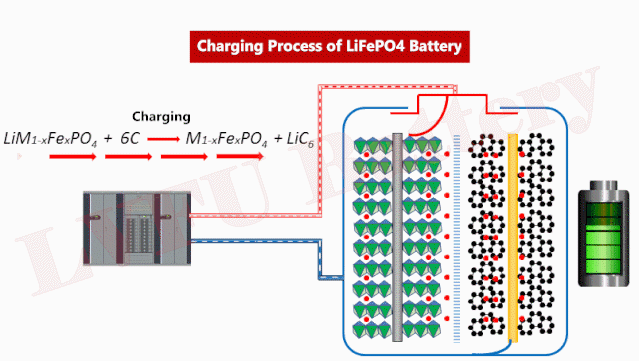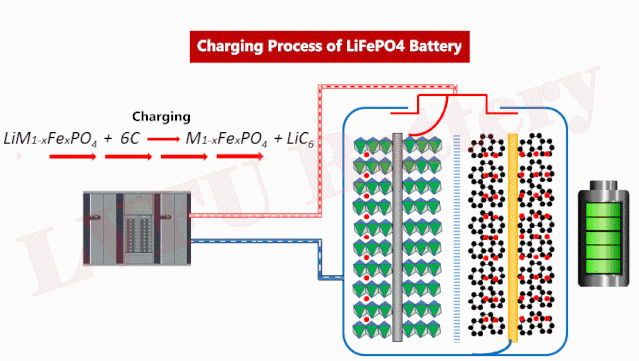
Iron phosphate is a black, water-insoluble compound with the formula LiFePO4. Compared to lithium-ion batteries, LFP batteries have several advantages. It has low manufacturing costs, long cycle life, and thermal stability. One drawback of LFP batteries is that they do not have the same energy and power density as those made with nickel-based positive electrodes. This means that EV batteries manufactured with LFP positive electrodes have shorter range and lower performance, but may be acceptable for low- and mid-priced EVs.
Key applications for LFPs also include energy storage systems and backup power supplies, whose low cost offsets concerns about reduced energy density.
Challenges in iron phosphate production
Iron phosphate is a relatively inexpensive and environmentally friendly material. The largest mining producers of phosphate ore are China, the United States, and Morocco. A huge new source was also discovered in Norway.
Iron phosphate is used industrially as a catalyst in the steel and glass industries and in the production of agricultural fertilizers. Abundant, the world’s reserves of phosphate rock are estimated to be sufficient for more than 100 years before it suddenly became popular as LFP traction batteries for EVs.
The increasing use of LFP batteries in electric vehicles and energy storage will require significantly more purified phosphoric acid (PPA). The automotive sector currently accounts for around 5% of purified phosphoric acid (PPA) demand, which is expected to jump to 24% by 2030.
Although the supply of mined phosphate material appears to be sufficient to meet future needs, refining the material into PPA does not meet demand. Currently, China dominates the phosphate refining industry, accounting for almost two-thirds of the production capacity. Currently, only three PPA refining projects are underway outside of China. A large-scale refinery capable of producing 30,000 tons of PPA will require a capital investment of $100 million, and many will need to be built by 2030 to meet the demand for increased LFP battery production. There is.
Refining phosphate rock to PPA must be done at very high levels for use in the positive electrode of LFP batteries. Unless heavy metals and impurities are removed, lithium ions can have difficulty migrating from the positive electrode (cathode) and negative electrode (anode). Currently, only about 3 percent of the total supply of phosphate minerals is available for refining into cathode battery materials.
It would also be beneficial to conduct PPA refining near the battery factories that use the material to make LFP cells. Transporting rocks containing phosphates is expensive, but refining the product closer to the battery factory reduces costs.Chinese companies such as BYD and CATL focus on the production of LFP batteries, so China has a big advantage in PPA refining.
LFP outlook
Beyond current LFP chemistry, the addition of manganese to the lithium iron phosphate cathode improves the energy density of the battery to a level close to that of nickel-based cathodes, thereby increasing the EV on a single charge. Cruising range increased. For these battery chemistries to continue to grow, significant investment in PPA refining capacity will be required, if LFP traction batteries are to be produced in large Western factories.