LiFePO4 Storage batteries are highly safe and have a service life 20 times longer than traditional batteries. They are a substitute for lead-acid batteries. In this blog post, we will share the battery production of LiFePO4 batteries so that users can understand its specific information from this aspect.
Chemical composition of battery production
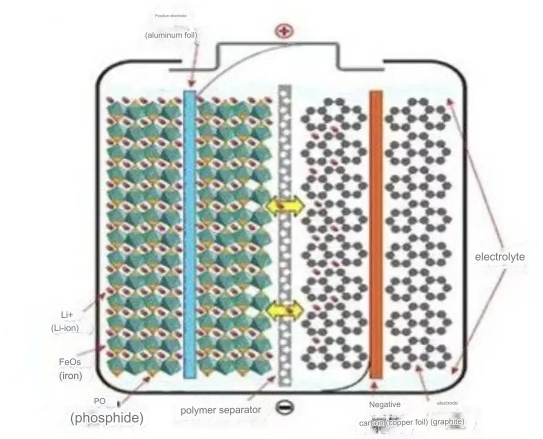
The battery production process of LiFePO4 Storage battery includes LiFePO4 as the positive electrode material, graphite, and liquid electrolyte as the harmful electrode material. These ingredients contribute to the battery’s electrochemical reactions during charge and discharge cycles. Liquid electrolytes are lithium salts dissolved in a solvent and play a crucial role in facilitating the movement of lithium ions between the anode and cathode. This ion transport determines its charge and discharge characteristics. LiFePO4 Storage batteries are also known for their excellent thermal stability, reduced risk of thermal runaway, and enhanced safety features.
Battery Production: The Process of Anode Manufacturing
The battery production of LiFePO4 Storage batteries requires high-purity raw materials for the positive electrode, including lithium carbonate and iron phosphate. A homogeneous mixture is obtained through a precise mixing process, ensuring uniform composition and optimal electrochemical performance. The next step is to apply the cathode material to the aluminum foil. This coating process requires the precise formation of a consistent coating that improves the cell’s overall efficiency. The coated foil is then dried and calendered to obtain the desired thickness and density.
Battery production: assembly and production of anodes
Similar to the positive electrode assembly, the anode material is applied to the current collector, and the coating must be applied evenly to optimize the electrochemical reactions within the cell. During the anode assembly process in battery production, adhesive materials often need to be introduced to enhance the adhesion between the anode material and the current collector. This step is critical to maintain structural integrity and prevent material from falling off, which could otherwise harm the battery’s performance. Anode assembly also involves the precise introduction of the electrolyte, which is critical to facilitating the movement of lithium ions between the anode and cathode during battery operation.
Battery assembly
First, the cathode and anode applied to the appliance are carefully aligned and placed between the cathode and anode. The next step is to inject the electrolyte into the battery. This liquid electrolyte fills the porous separator, creating channels for lithium ions to move during battery operation. During this step, avoid air bubbles and ensure even electrolyte distribution. Once the components are in place, they are sealed to prevent leaks and protect the internal components. Automated production lines are often used throughout the assembly process to increase efficiency and consistency.
Quality control measures
The last critical step in battery production is quality inspection. After the LiFePO4 Storage battery is assembled, the battery undergoes comprehensive off-line testing. Performance and safety need to be verified through more rigorous evaluation. Batteries that do not meet strict standards will be rejected to maintain the highest quality standards. Each battery is tracked throughout the production process through an advanced traceability system. This ensures that any issues can be traced back to their root cause, promoting continuous improvement and corrective action.
Visual battery production
The production technology of LiFePO4 Storage batteries includes chemistry, battery assembly, and quality control. Through the popular science articles about cathode and anode production, electrolyte formula and other related information, users can deeply understand the process of these advanced energy storage solutions.